Radioactive and Stable Isotopes
Concept Question: What is the difference between a radioactive isotope and a stable isotope?
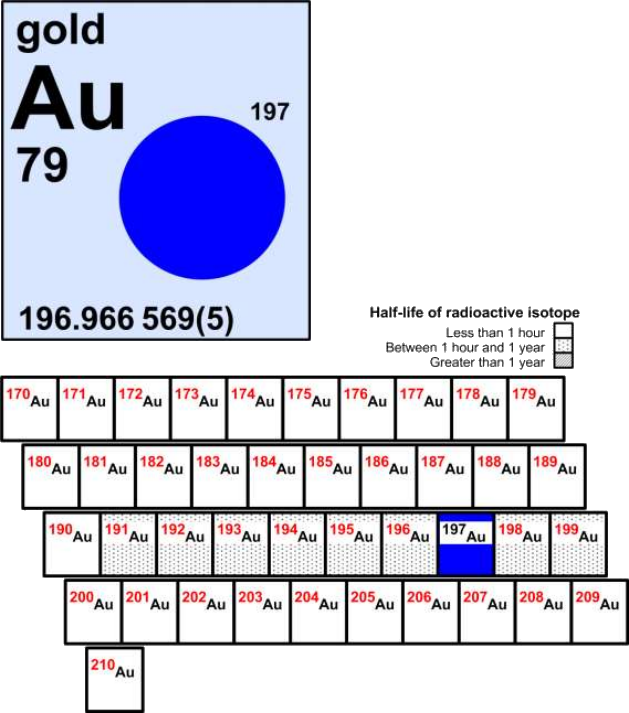
There are two types of isotopes: stable and radioactive. Let's look at gold for an example. Gold has 41 known isotopes, ranging from gold-170 to gold-210. Only one of these, gold-197, is stable, the rest are radioactive.
A stable isotope has no experimentally detected nuclear decays. A decay event happens when there is a spontaneous transformation within the nucleus of an atom, changing one isotope to another isotope, or even to another element. Isotopes which undergo these decays are referred to as radioactive, or radioisotopes.
One of the ways we can measure radioactivity is through half-lives. A radioactive half-life is defined as the amount of time it takes for half of a sample of a radioactive isotope to undergo a nuclear decay event. This means that if you have a sample of a radioactive substance with a half-life of 1 day, only half of your sample will be of the original substance after 1 day. The rest of it may become different isotopes of the same element, or atoms a different element.
IUPAC sorts elements into one of three categories based on their half-lives: less than 1 hour, between 1 hour and 1 year, and greater than 1 year. Open the IUPAC Periodic Table of the Elements and Isotopes Learning Tool and click on gold. Select the 'more information' link and read through the document. What kind of technologies can radioactive gold isotopes be used for?